CT Chest (Thorax) - CAM 750
GENERAL INFORMATION
It is an expectation that all patients receive care/services from a licensed clinician. All appropriate supporting documentation, including recent pertinent office visit notes, laboratory data, and results of any special testing must be provided. If applicable: All prior relevant imaging results and the reason that alternative imaging cannot be performed must be included in the documentation submitted.
Where a specific clinical indication is not directly addressed in this guideline, medical necessity determination will be made based on widely accepted standard of care criteria. These criteria are supported by evidence-based or peer-reviewed sources such as medical literature, societal guidelines and state/national recommendations.
Policy
Chest Computed Tomography (CT) generates images of the organs and structures in the chest (thorax) with the use of radiation.
INDICATIONS FOR CHEST CT
Screening for Lung Cancer
- The use of low-dose, non-contrast spiral (helical) multi-detector CT imaging as an annual screening technique for lung cancer is considered medically necessary ONLY when used to screen for lung cancer for certain high-risk asymptomatic individuals when ALL of the following criteria are met:1
- Individual is between 50 – 80 years of age*; AND
- There is at least a 20 pack-year history of cigarette** smoking
*May approve for individuals over the age limit if the individual is a candidate for and willing to undergo curative treatment upon diagnosis
**Only personal cigarette smoking history as above places an individual at high risk; secondhand smoke exposure and other forms of smoking (such as pipe, cigar, marijuana, vaping) do NOT factor into current recommendations for LDCT Screening.
- Screening should be discontinued once a person develops a health problem that limits the willingness or ability to have curative intent treatment.2,3
Follow-Up of Lung Nodules
Lung Nodules With No History of Malignancy4
- Incidental pulmonary nodule on X-ray:
- Immediate follow up with Chest CT if the nodule is indeterminate as per the radiology report (i.e. not typical of granulomatous disease)
- Incidental pulmonary nodules on non-chest CT (such as a neck, shoulder or abdomen CT):
- Nodules > 8 mm or those with very suspicious features need a dedicated Chest CT as early as possible
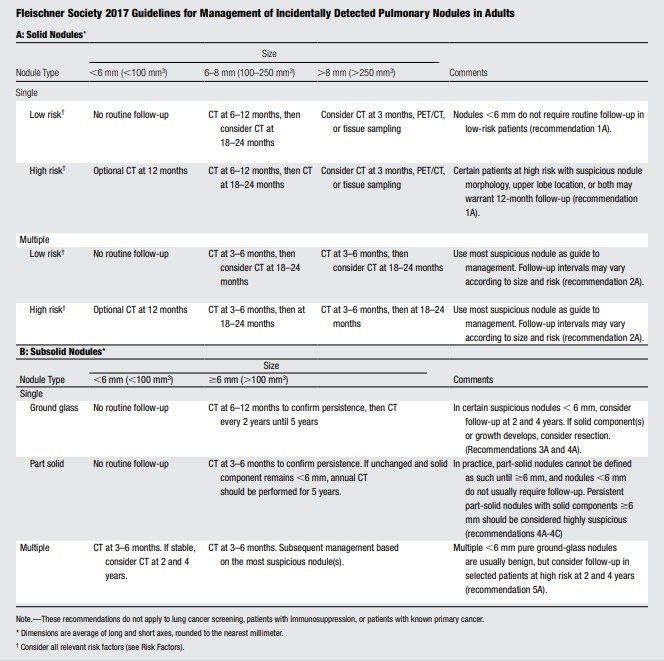
- Nodules ≤ 8 mm should follow the Fleischner table (Table 2) to determine when a dedicated Chest CT is needed
- Follow-up of pulmonary nodules detected on a non-screening (regular) Chest CT is as per Fleischner table (see Table 2) when ALL of the following criteria are met:
- Individual is ≥ 35 years old.
- If age is < 35, non-malignant causes are more likely and infection should be excluded. Once infection is excluded if the nodule is unable to followed with chest X-ray and explanation given as to how management would change, coverage can be considered on a case-by-case basis.
- No personal history of cancer
- If there is a prior history of cancer, follow-up imaging can be done as soon as 3 months.
- Not immunocompromised
- If the patient is immunocompromised, follow-up imaging can be done as soon as 1 month if there is suspicion of fulminant infection.
- Patient does not qualify for screening with LDCT nor was the nodule was not identified on LDCT (uses Lung-Rads criteria rather than the Fleischner table).
- Individual is ≥ 35 years old.
NOTE: If multiple nodules, the largest and type is used for decision.
Follow-Up of Lung Nodule Seen on Screening LDCT
- Follow-up of pulmonary nodule identified on screening LDCT (for patients at high risk for lung cancer) as per Lung-Rads criteria5,6 (see Table 1 — typically ordered as a LDCT)
NOTE: If multiple nodules, the largest and type is used for decision.
Infection and Inflammation
Infection Follow-Up Imaging
- Abscess, empyema, or pleural effusions on chest X-ray7
- For evaluation of non-resolving pneumonia or inflammatory disease documented by at least two imaging studies:
- Unimproved with 4 weeks of antibiotic treatment; OR
- Unresolved at 8 weeks8
Interstitial Lung Disease
- For evaluation of suspected ILD after initial chest x-ray excludes a more acute disease (such as pneumonia) in the following situations:9
- Based on restrictive pattern pulmonary function test
- In patients with known collagen vascular disease in whom ILD is suspected
- For biopsy guidance when ILD is suspected10
- With signs or symptoms unresponsive to treatment such as:
- Shortness of breath
- Persistent dyspnea
- Persistent cough
- For reassessment of known ILD
- annually AND
- for clinical progression of disease
NOTE: Chest CT for evaluation of ILD is typically performed with a high-resolution protocol (HRCT)
Sarcoidosis11
- For suspected sarcoidosis after initial workup including chest X-ray
- For known sarcoidosis when there are EITHER symptoms of progression (including normal CXR with unexplained dyspnea or cough) OR progression on chest X-ray
Granulomatosis with Polyangiitis (Wegener's Granulomatosis)
- For evaluation of suspected granulomatosis with polyangiitis (Wegener's Granulomatosis) after initial workup including labs and chest X-ray suggest the diagnosis
- For follow-up of lesions after treatment12
Tuberculosis (TB)13,14
- Known or suspected tuberculosis and initial chest X-ray completed
Covid-19 (Coronavirus)
- Known acute COVID AND worsening respiratory status AND indeterminate chest X-ray
- Known long (chronic) COVID AND EITHER:15
- Restricted diffusion on pulmonary function test OR
- Low oxygen saturation and prior chest X-ray OR
- Known fibrosis with persistent symptoms
Chronic Cough (16)
- Symptoms > 8 weeks and chest X-ray completed and EITHER
- Clinical concern for bronchiectasis OR
- After evaluation for other causes and failed treatment for those diagnosed with:
- Asthma
- Gastroesophageal Reflux Disease
- Discontinuation of ACE inhibitors
- Postnasal drip
Thoracic Aortic Disease17,18,19,20,21
Suspected Thoracic Aortic Aneurysm (TAA)
- Asymptomatic suspected thoracic aortic aneurysm
- Based on other imaging such as echocardiogram or chest X-ray
- Screening in individuals with a personal history of bicuspid aortic valve when TTE (transthoracic echocardiogram) is inconclusive or insufficient:20
- Baseline study at diagnosis
- Every 3 years thereafter
- Screening in individuals at elevated risk due to family history as below when TTE (transthoracic echocardiogram) is inconclusive or insufficient:20
- First-degree relatives of individuals with a known thoracic aortic aneurysm (defined as > 50% above normal) or dissection
- First and second-degree relatives of individuals with familial thoracic aortic aneurysm and dissection (FTAAD)/nonsyndromic heritable thoracic aortic disease (NS-TAD)
- First degree relatives of individuals with a known bicuspid aortic valve
- Symptomatic known or suspected thoracic aortic aneurysm
- Signs and Symptoms may include:
- Abrupt onset of severe sharp or stabbing pain in the chest, back or abdomen
- Asymmetric blood pressure between limbs
- Acute chest or back pain and at high risk for aortic aneurysm and/or aortic syndrome (risk factors include hypertension, atherosclerosis, prior cardiac or aortic surgery, underlying aneurysm, connective tissue disorder (e.g., Marfan syndrome, vascular form of Ehlers-Danlos syndrome, Loeys-Dietz syndrome), and bicuspid aortic valve)20,22
- Signs and Symptoms may include:
- Suspected vascular cause of dysphagia or expiratory wheezing with other imaging that is suggestive or inconclusive
Thoracic Aortic Syndromes
- For suspected acute aortic syndrome (AAS) such as aortic dissection, intramural hematoma and penetrating atherosclerotic ulcer:
- Other imaging (such as echocardiogram) is suggestive of AAS OR
- Individual is either:
- High risk and one sign/symptom OR non-high risk and two or more signs/symptoms of AAS:
- High risk conditions:
- Marfan's syndrome or other connective tissue disease, family history of aortic disease, known aortic valve disease, recent aortic manipulation and/or known thoracic aortic aneurysm
- High risk conditions:
- High risk and one sign/symptom OR non-high risk and two or more signs/symptoms of AAS:
- Signs and symptoms concerning for AAS:
- Chest, back or abdominal pain described as abrupt onset, severe in intensity and/or ripping or tearing in quality
- Pulse deficit or systolic blood pressure differential
- Focal neurologic deficit with pain
- Hypotension or shock
- For follow-up of known aortic syndromes, including aortic dissection, intramural hematoma and penetrating atherosclerotic ulcer: frequency for follow up is as clinically indicated
Follow-Up of Known Thoracic Aortic Aneurysm
- Baseline study at diagnosis then every 6 – 24 months
- If there is a change in clinical status or cardiac exam, then imaging sooner than 6 months is indicated
Postoperative Follow-Up of Aortic Repair
- Follow-up thoracic endovascular aortic repair (TEVAR):20
- Baseline post-EVAR at 1 month post-EVAR
- Annually thereafter if stable
- More frequent imaging (as clinically indicated) may be needed if there are complications or abnormal findings on surveillance imaging
- Follow up open repair at the following intervals:
- Baseline follow-up study at one year post-operatively
- Every 5 years thereafter
- If abnormal findings are seen on any surveillance imaging study, imaging is then done annually
Non-Aortic Vascular Disease
- Superior Vena Cava (SVC) syndrome23
- SVC syndrome is a clinical diagnosis and may be suspected when there are signs of venous congestion in the upper body (such as shortness of breath, distended neck veins and facial/upper extremity edema)
- Thoracic outlet syndrome
- Suspected pulmonary hypertension when other testing (echocardiogram or right heart catheterization) is suggestive of the diagnosis24
NOTE: Chest CT is NOT approvable for pulmonary embolism (PE). See CAM 749, Chest CTA.
Congenital Malformation
- Thoracic malformation on chest X-ray19
- Congenital Heart Disease with pulmonary hypertension25
- Malformations (such as pectus excavatum, pectus carinatum, scoliosis) in patients with cardiorespiratory symptoms for whom treatment is being considered
Transplants
Pre-Transplant
- Prior to solid organ transplantation (donor or recipient)
- For workup prior to bone marrow transplant (BMT)
Post-Transplant26
- Routine surveillance of prior lung transplantation
- Concern for complication at any time following lung transplantation (CXR not required)
Chest Wall Pain and Injuries
- Non-traumatic chest wall pain with normal chest X-ray and or rib X-ray with EITHER:27
- Suspicion of malignancy OR
- Signs and symptoms of infection, such as: fever, elevated inflammatory markers, known infection at other sites OR
- History of chest radiation or chest surgery
- Suspected chest wall injuries (including musculotendinous, costochondral cartilage, sternoclavicular joint, and manubriosternal joint injuries), when imaging will potentially alter management
Other Indications
- Pneumothorax on chest X-ray when imaging findings will change management
- Hemoptysis after X-ray completed28
- Vocal cord paralysis on endoscopic exam
- Phrenic nerve paralysis on diaphragm fluoroscopy (fluoroscopic) sniff test
Suspected Malignancy
- Non-lung parenchymal29
- Mass or lesion, including lymphadenopathy, after inconclusive initial imaging:
- Can allow one follow-up to ensure stability
- Additional follow-up may be approved if changing on repeat imaging
- Mass or lesion, including lymphadenopathy, after inconclusive initial imaging:
- Chest wall
- Mass or lesion after inconclusive initial imaging when MRI is contraindicated or cannot be performed
- For further evaluation of the following:
- Weight loss of ≥ 5% over 12 months AND signs and symptoms consistent with a source in the chest (such as a smoker with a cough) after initial chest X-ray
- Weight loss of ≥ 5% over 12 months when initial evaluation with Chest X-ray, age-appropriate cancer screening (such as colonoscopy and mammography), labs (including CBC, CMP, HbA1C, TSH, stool hemoccult, ESR/CRP, HIV, Hepatitis C) fail to identify a cause AND there is documented further decline in weight
- Gestational trophoblastic disease when hCG fails to decline appropriately following surgery
- Suspected paraneoplastic syndrome (including dermatomyositis) when appropriate workup has been done and there is a suspicion of malignancy
- Thymoma screening in Myasthenia Gravis patients30
Known Malignancy
Initial Staging31,32,33,34
- Chest CT is appropriate for initial staging of the majority of malignancies when either biopsy proven or suspected based on prior imaging.
Restaging
- Chest CT is indicated for restaging during active treatment (every 2 – 3 cycles of chemo or immunotherapy, following radiation and/or after surgery) for the majority of cancers except the following:
- Bladder cancer (non-muscle invasive)
- Breast cancer (stage I – III)
- Colon cancer (stage I)
- Prostate cancer (non-metastatic)
- The above excluded malignancies would require initial evaluation (such as Chest x-ray) with findings concerning for chest pathology.
- Chest CT is indicated in addition to PET while on active treatment every 2 – 3 cycles of chemo or immunotherapy for the following: Ewing Sarcoma, Osteosarcoma, Hodgkin Lymphoma, Pediatric Aggressive Mature B-Cell Lymphomas, Pediatric Hodgkin Lymphoma, Soft Tissue Sarcoma (if receiving systemic chemotherapy)
Surveillance
Chest CT is indicated during surveillance for the following malignancies at the intervals defined below:
- Adrenocortical carcinoma: every 3 – 12 months for 5 years then as clinically indicated
- Anal carcinoma: every 3-6 months for 1-2 years, then every 6 – 12 months for an additional year
- Biliary tract cancers (ampullary adenocarcinoma, cholangiocarcinomas and gallbladder): every 3 – 6 months for 2 years then every 6 – 12 months for up to 5 years then as clinically indicated
- Bone tumors and sarcomas (chondrosarcoma, chordoma, giant cell tumor of bone, Ewing sarcoma, soft tissue sarcoma, osteosarcoma)
- Every 3 – 6 months for 5 years, then annually for and additional 5 years then as clinically indicated
- Bladder cancer (muscle Invasive only): every 3 – 6 months for 2 years, then annually for up to 5 years then as clinically indicated
- Colon cancer (Stage II or higher): every 6 – 12 months for 5 years, then as clinically indicated
- Esophageal and esophagogastric junction cancers: every 3 – 6 months for 2 years, then annually for up to 5 years
- Gastric cancer: every 6 months for 2 years, then annually up to 5 years
- Hepatocellular carcinoma: every 3 – 6 months for 2 years, then every 6 months indefinitely
- Lymphoma (follicular, diffuse large B-cell, Burkitt, Hodgkin, marginal zone, T-cell) and hairy cell leukemia:
- Every 3 – 6 months for 2 years, then annually
- Melanoma (stage II or higher): every 3 – 12 months for 2 years then every 6 – 12 months for 3 years, then as clinically indicated
- Merkel cell carcinoma: every 3 – 6 months for 3 years, then every 6 – 12 months indefinitely
- Mesothelioma (pleural and peritoneal): every 3 – 6 months for 5 years, then annually until 10 years, then as clinically indicated
- Neuroblastoma: every 3 months for 1 year, then every 6 – 12 months for 2 years, then as clinically indicated
- Neuroendocrine tumors: every 3 – 6 months for 5 years, then every 6 – 12 months for 5 years, then as clinically indicated
- Non-small cell lung cancer: every 3 months for 3 years, then every 6 months for 2 years, then annually
- Occult primary tumors: every 3 – 6 months for 2 years, then every 6 – 12 months for 3 years, then annually
- Ovarian cancer: every 3 – 6 months for 2 years, then every 6 – 12 months for 3 years, then as clinically indicated
- Pancreatic cancer: every 3 – 6 months for 2 years, then every 6 – 12 months as clinically indicated
- Penile cancer: Every 3 – 6 months for 2 years, then every 6 – 12 months for an additional 3 years, then as clinically indicated
- Prostate cancer (observation): as clinically indicated for rising PSA or symptoms suggestive of progression
- Renal cell carcinoma:
- Stage I — annually for 5 years, then as clinically indicated
- Stage II and higher — every 3 – 6 months for 3 years, then annually for 2 years, then as clinically indicated
- Rectal cancer:
- Stage II, III — every 6 – 12 months for 5 years, then as clinically indicated
- Stage IV – every 3 – 6 months for 2 years, every 6 – 12 months for a total of 5 years
- Small bowel adenocarcinoma: every 6 – 12 months for 5 years
- Small cell lung cancer: every 2 months for the first year, every 3 – 4 months for years 2 and 3, then every 6 months during years 4 and 5 then annually
- Testicular cancer (Stage IIA and higher): every 3 months for 1 year, then every 6 months for 1 year then annually for 2 years
- Thymoma and thymic carcinoma: every 3 – 6 months for 2 years, then annually for up to 5 years, then as clinically indicated
- Urothelial carcinoma of the prostate, primary carcinoma of the urethra: high-risk patients only: every 3 – 6 months for 2 years, then annually
- Uveal melanoma: every 6 – 12 months for 10 years, then as clinically indicated
- Wilm's tumor: every 3 months for 2 years, then every 6 months for 2 years
NOTE: For any patient with stage IV cancer (any type) that is either in remission or on a treatment break, chest CT is indicated every 3 – 6 months.
When a cancer is not listed above, chest CT is not routinely a part of surveillance for that cancer in an asymptomatic patient. There would need to be a sign or symptom of recurrence to consider chest CT.
When the timeframe above for routine surveillance has elapsed, there would need to be a sign or symptom of recurrence to consider chest CT.
Preoperative/procedural evaluation
- Pre-operative evaluation for a planned surgery or procedure
- Pre-operative evaluation for electromagnetic navigation bronchoscopy35 (This is a non-diagnostic CT.)
Postoperative/procedural evaluation
- Post-surgical follow-up when records document medical reason requiring additional imaging
Further Evaluation of Indeterminate Findings on Prior Imaging
Unless follow-up is specified within the guideline:
- For initial evaluation of an inconclusive finding on a prior imaging report that requires further clarification
- One follow-up exam of a prior indeterminate MR/CT finding to ensure no suspicious interval change has occurred. (No further surveillance unless specified as highly suspicious or change was found on last follow-up exam.)
Genetic Syndromes and Rare Diseases
- Cystic fibrosis: chest CT every 2 years and as needed to assess for bronchiectasis
- Multiple endocrine neoplasia Type 1 (MEN1): annually36,37
- Hereditary paraganglioma-pheochromocytoma (PGL/PCC) syndrome (SDHx mutations): every 2 years when whole-body MRI (CPT 76498) is not available
- For other syndromes and rare diseases not otherwise addressed in the guideline, coverage is based on a case-by-case basis using societal guidance.
Combination Studies
Chest/Abdomen and Pelvis CT
- As numerous disease processes, including but not limited to malignancy, may affect the chest, abdomen and pelvis, this combination is indicated when the guideline criteria for BOTH Chest CT and Abdomen and Pelvis CT have been met.
Chest CT/Abdomen and Pelvis CT and PET
- CT of the original sites of disease is indicated in addition to PET while on active treatment every 2 – 3 cycles of chemo or immunotherapy for the following: Hodgkin lymphoma, pediatric aggressive mature B-cell lymphomas, pediatric Hodgkin lymphoma.
Chest CT and Brain/Abdomen/Pelvis MRI
- Multiple endocrine neoplasia syndrome Type 1 (MEN-1)
- Chest/abdomen/pelvis annually
- Brain/chest/abdomen/pelvis every 3 years
Chest CT and PET
- Chest CT is indicated in addition to PET while on active treatment every 2 – 3 cycles of chemo or immunotherapy for the following: Ewing sarcoma, osteosarcoma, soft tissue sarcoma (if receiving systemic chemotherapy).
Chest CTA (or MRA) and Chest CT (or MRI)
- When needed for clarification of vascular invasion from tumor
Neck/Abdomen/Pelvis MRI and Chest CT
- PGL/PCC (hereditary paraganglioma/pheochromocytoma syndromes or SDHx mutations): every 2 years IF whole body MRI (unlisted MRI CPT 76498) is not available38 (See Unlisted Studies Evolent_CG_063.)39
Neck/Chest CT
- Vocal cord paralysis on endoscopic exam and concern for recurrent laryngeal nerve lesion
Neck/Chest CT
- Vocal cord paralysis on endoscopic exam and concern for recurrent laryngeal nerve lesion
- Phrenic nerve paralysis on diaphragm fluoroscopy (fluoroscopic sniff test)
Sinus CT/Chest CT
- Granulomatosis with polyangiitis (Wegener’s granulomatosis) disease (GPA)
Sinus/Chest/Abdomen/Pelvis CT and Brain MRI
- Prior to bone marrow transplant
Combination Studies for Malignancy for Initial Staging or Restaging
Unless otherwise specified in this guideline, indication for combination studies for malignancy for initial staging or restaging:
- Concurrent studies to include CT or MRI of any of the following areas as appropriate depending on the cancer: Brain, Neck, Chest, Abdomen, Pelvis, Cervical Spine, Thoracic Spine or Lumbar Spine
Rationale/Background
Fever of Unknown Origin
Initial workup prior to CT would include a comprehensive history, repeated physical exam, complete blood count with differential, three sets of blood cultures, chest X-ray, complete metabolic panel, urinalysis, ESR, ANA, RA, CMV IgM antibodies, virus detection in blood, heterophile antibody test, tuberculin test, and HIV antibody test.61 Lastly, with a negative CXR, only when initial workup and abdomen/pelvis CT/MR fail to identify the cause for fever can chest CT be approved. If CXR suggests a malignancy and/or source of fever, then chest CT would be approved.
Suspected paraneoplastic syndromes with no established cancer diagnosis: laboratory evaluation and imaging
The laboratory evaluation for paraneoplastic syndrome is complex. If the appropriate lab test results are suspicious for malignancy, imaging is indicated.
For SIADH (hyponatremia + increased urine osmolality), there is a high association with small cell lung cancer, therefore imaging typically starts with chest CT. If other symptoms suggest a different diagnosis other than small cell lung cancer, different imaging studies may be reasonable.
For hypercalcemia (high serum calcium, low-normal PTH, high PTHrP) it is reasonable to start with bone imaging followed by a more directed evaluation such as mammogram, chest, abdomen and pelvis imaging as appropriate.
For Cushing syndrome (hypokalemia, normal-high midnight serum ACTH NOT suppressed with dexamethasone) abdominal and chest imaging is reasonable. If dexamethasone suppression test DOES suppress ACTH, pituitary MRI is reasonable.
For hypoglycemia, labs drawn during a period of hypoglycemia (glucose < 55, typically a 72 hour fast) (insulin level, C-peptide and IGF-2:IGF-1 ratio) should be done to evaluate for an insulinoma. An elevated insulin level, elevated C-peptide and/or normal IGF-2:IGF-1 ratio warrant CT or MRI abdomen to look for insulinoma. A low insulin, low C-peptide and/or elevated IGF-2:IGF-1 ratio warrant chest and abdominal imaging.
When a paraneoplastic neurologic syndrome is suspected, nuclear and cytoplasmic antibody panels are often ordered to further identify specific tumor types. Results are needed prior to imaging. Because these tests are highly specific, if an antibody highly associated with a specific cancer is positive, then further imaging for that cancer is reasonable. For example, anti-Hu has a high association with SCLC and chest CT would be reasonable. Anti-MA2 has a high association with testicular cancer and testicular ultrasound would be a reasonable next step.
Table 1: Lung-RADS Assessment Categories
*This table is reproduced without alteration or edit in accordance with provisions in a Creative Commons License. The full document and license information can be found here: Lung Rads | American College of Radiology (acr.org)
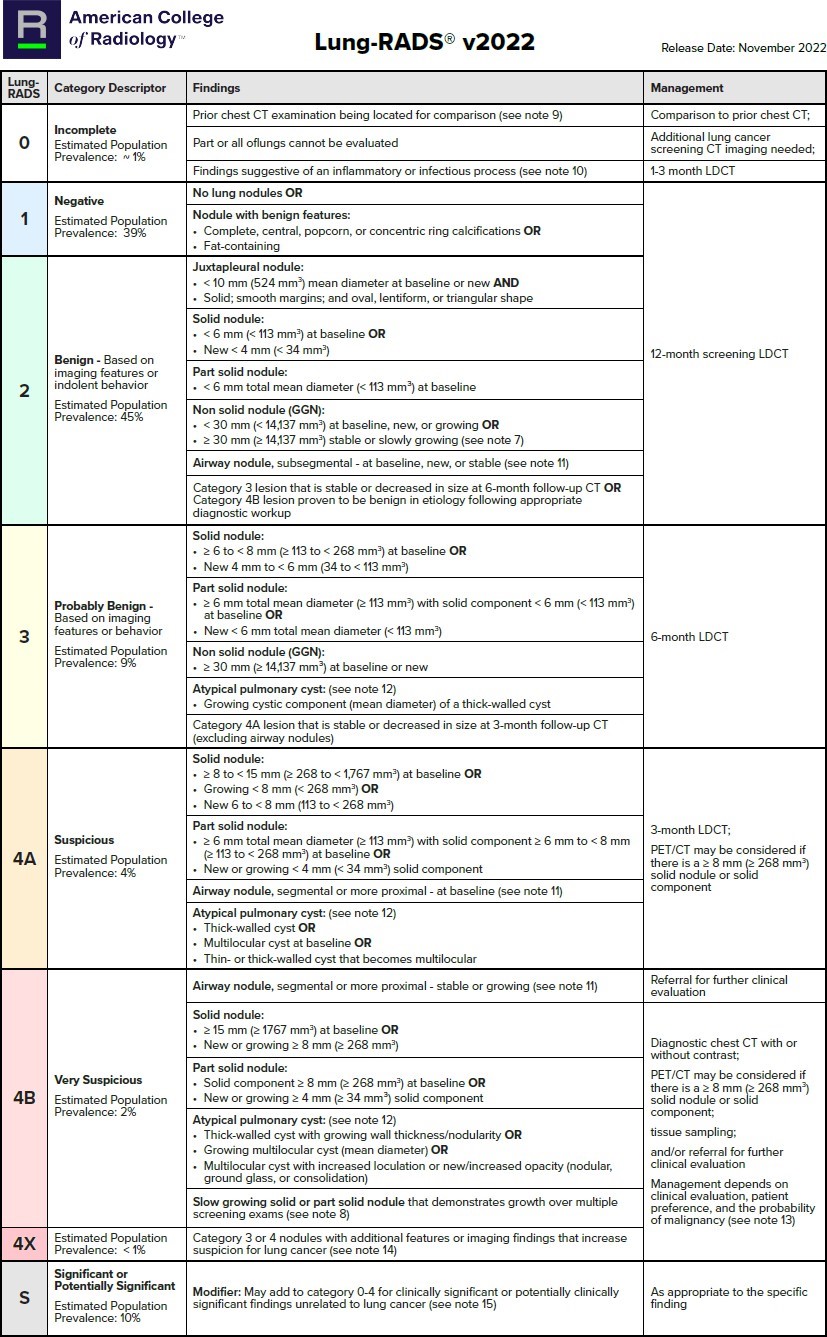
Table 2: 2017 Fleischner Society Guidelines for Management of Incidental Detected Pulmonary Nodules (40)
Fleischner Table
*This table is reproduced without alteration or edit.
Contraindications and Preferred Studies
- Contraindications and reasons why a CT/CTA cannot be performed may include impaired renal function, significant allergy to IV contrast, pregnancy (depending on trimester).
- Contraindications and reasons why an MRI/MRA cannot be performed may include impaired renal function, claustrophobia, non-MRI compatible devices (such as non- compatible defibrillator or pacemaker), metallic fragments in a high-risk location, patient exceeds weight limit/dimensions of MRI machine.
References
- Final Recommendation Statement Lung Cancer: Screening. March 9, 2021.
- NCCN Clinical Practice Guidelines in Oncology: Lung Cancer Screening v2.2024. J Natl Compr Canc Netw. 2024.
- Wolf A, Oeffinger K, Shih T, Walter L, Church T et al. Screening for lung cancer: 2023 guideline update from the American Cancer Society. CA Cancer J Clin. 2024; 74: 50 - 81. https://doi.org/10.3322/caac.21811.
- Bueno J, Landeras L, Chung J. Updated Fleischner Society Guidelines for Managing Incidental Pulmonary Nodules: Common Questions and Challenging Scenarios. Radiographics. Sep-Oct 2018; 38: 1337-1350. 10.1148/rg.2018180017.
- McKee B, Regis S, McKee A, Flacke S, Wald C. Performance of ACR Lung-RADS in a clinical CT lung screening program. J Am Coll Radiol. Mar 2015; 12: 273-6. 10.1016/j.jacr.2014.08.004.
- Pinsky P, Gierada D, Black W, Munden R, Nath H et al. Performance of Lung-RADS in the National Lung Screening Trial: a retrospective assessment. Ann Intern Med. Apr 7, 2015; 162: 485-91. 10.7326/m14-2086.
- Zaki H, Albaroudi B, Shaban E, Shaban A, Elgassim M et al. Advancement in pleura effusion diagnosis: a systematic review and meta-analysis of point-of-care ultrasound versus radiographic thoracic imaging. The Ultrasound Journal. 2024; 16: true. 10.1186/s13089-023-00356-z.
- Little B, Gilman M, Humphrey K, Alkasab T, Gibbons F et al. Outcome of recommendations for radiographic follow-up of pneumonia on outpatient chest radiography. AJR Am J Roentgenol. Jan 2014; 202: 54-9. 10.2214/ajr.13.10888.
- American College of Radiology. ACR Appropriateness Criteria® Diffuse Lung Disease. American College of Radiology. 2021; https://acsearch.acr.org/docs/3157911/Narrative/.
- ACR-STR. ACR-STR Practice Parameter for the Performance of High-Resolution Computed Tomography (HRCT) of the Lungs in Adults. American College of Radiology. 2020.
- Bokhari S, Zulfiqar H, Mansur A. Sarcoidosis. [Updated 2023 Jun 25]. StatPearls [Internet]. 2023; https://www.ncbi.nlm.nih.gov/books/NBK430687/.
- Lassandro G, Picchi S G, Corvino A, Massimo C, Tamburrini S et al. Noninfectious Granulomatous Lung Disease: Radiological Findings and Differential Diagnosis. Journal of Personalized Medicine. 2024; 14: 10.3390/jpm14020134.
- 13. American College of Radiology. ACR Appropriateness Criteria® Imaging of Possible Tuberculosis. 2016.
- Ko Y, Lee H, Park Y, Hong S, Shin J et al. Correlation of microbiological yield with radiographic activity on chest computed tomography in cases of suspected pulmonary tuberculosis. PLoS One. 2018; 13: e0201748. 10.1371/journal.pone.0201748.
- Rubin G, Ryerson C, Haramati L, Sverzellati N, Kanne J et al. The Role of Chest Imaging in Patient Management during the COVID-19 Pandemic: A Multinational Consensus Statement from the Fleischner Society. Radiology. Jul 2020; 296: 172-180. 10.1148/radiol.2020201365.
- Turner R, Bothamley G. Chronic cough and a normal chest X-ray - a simple systematic approach to exclude common causes before referral to secondary care: a retrospective cohort study. NPJ Prim Care Respir Med. Mar 3, 2016; 26: 15081. 10.1038/npjpcrm.2015.81.
- Borger M A, Fedak P W M, Stephens E H, Gleason T G, Girdauskas E et al. The American Association for Thoracic Surgery consensus guidelines on bicuspid aortic valve-related aortopathy: Full online-only version. J Thorac Cardiovasc Surg. 2018; 156: e41-e74. 10.1016/j.jtcvs.2018.02.115.
- Erbel R, Aboyans V, Boileau C, Bossone E, Bartolomeo R D. 2014 ESC Guidelines on the diagnosis and treatment of aortic diseases: Document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. The Task Force for the Diagnosis and Treatment of Aortic Diseases of the European Society of Cardiology (ESC). Eur Heart J. 2014; 35: 2873-926. 10.1093/eurheartj/ehu281.
- Ferreira Tda A, Chagas I, Ramos R, Souza E. Congenital thoracic malformations in pediatric patients: two decades of experience. J Bras Pneumol. Mar-Apr 2015; 41: 196-9. 10.1590/s1806- 37132015000004374.
- Isselbacher E, Preventza O, Hamilton Black III J, Augoustides J, Beck A et al. 2022 ACC/AHA Guideline for the Diagnosis and Management of Aortic Disease: A Report of the American Heart Association/American College of Cardiology Joint Committee on Clinical Practice Guidelines. Journal of the American College of Cardiology. 2022; 80: e223 - e393. https://doi.org/10.1016/j.jacc.2022.08.004.
- Mariscalco G, Debiec R, Elefteriades J, Samani N, Murphy G. Systematic Review of Studies That Have Evaluated Screening Tests in Relatives of Patients Affected by Nonsyndromic Thoracic Aortic Disease. Journal of the American Heart Association. 2018; 7: true. 10.1161/JAHA.118.009302.
- Murillo H, Molvin L, Chin A S, Fleischmann D. Aortic Dissection and Other Acute Aortic Syndromes: Diagnostic Imaging Findings. Radiographics: a review publication of the Radiological Society of North. 2021; 41: 425-446.
- Seligson M, Surowiec S. Superior Vena Cava Syndrome. [Updated 2022 Sep 26]. StatPearls [Internet]. 2022; Accessed: May 2024. https://www.ncbi.nlm.nih.gov/books/NBK441981/.
- Chen R, Liao H, Deng Z, He Z, Zheng Z et al. Efficacy of computed tomography in diagnosing pulmonary hypertension: A systematic review and meta-analysis. Front Cardiovasc Med. 2022; 9: 966257. 10.3389/fcvm.2022.966257.
- Pascall E, Tulloh R. Pulmonary hypertension in congenital heart disease. Future Cardiol. Jul 2018; 14: 343-353. 10.2217/fca-2017-0065.
- Kim S J, Azour L, Hutchinson B D, Shirsat H, Zhou F et al. Imaging Course of Lung Transplantation: From Patient Selection to Postoperative Complications. RadioGraphics. 2021; 41: 1043-1063. 10.1148/rg.2021200173.
- American College of Radiology. ACR Appropriateness Criteria: Nontraumatic Chest Wall Pain. Journal of the American College of Radiology. 2021; 18: S394 - S405. 10.1016/j.jacr.2021.08.004.
- American College of Radiology. ACR Appropriateness Criteria® Hemoptysis. 2019; 2022:
- Mansour J, Raptis D, Bhalla S, Heeger A, Abbott G et al. Diagnostic and Imaging Approaches to Chest Wall Lesions. RadioGraphics. 2022; 42: 359 - 378. 10.1148/rg.210095.
- Kumar R. Myasthenia gravis and thymic neoplasms: A brief review. World J Clin Cases. Dec 16, 2015; 3: 980-3. 10.12998/wjcc.v3.i12.980.
- Diessner J, Anders L, Herbert S, Kiesel M, Bley T et al. Evaluation of different imaging modalities for axillary lymph node staging in breast cancer patients to provide a personalized and optimized therapy algorithm. Journal of Cancer Research and Clinical Oncology. 2023; 149: 3457 - 3467. 10.1007/s00432-022-04221-9.
- Erasmus L, Strange T, Agrawal R, Strange C, Ahuja J et al. Lung Cancer Staging: Imaging and Potential Pitfalls. 2023; 13: 10.3390/diagnostics13213359.
- Feng S, Yang S. The new 8th TNM staging system of lung cancer and its potential imaging. Diagnostic and interventional radiology (Ankara, Turkey). 2019; 25: 270-279.
- Schmidlin E, Gill R. New frontiers in esophageal radiology. Annals of translational medicine. 2021; 9: 904.
- Fu Y, Yuan X, Pan W, Lu Y, Zeng D. Comparative analysis of the efficacy and safety of electromagnetic navigation bronchoscopy combined with x-ray or radial endobronchial ultrasound biopsy in the diagnosis of small peripheral pulmonary nodules. Thorac Cancer. 2023; 14: 1348 - 1354. https://doi.org/10.1111/1759-7714.14879.
- NCCN. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) Genetic/Familial High-Risk Assessment: Breast, Ovarian, and Pancreatic v3.2024. National Comprehensive Cancer Network®. 2024.
- NCCN. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) Neuroendocrine and Adrenal Tumors Version 1.2023. National Comprehensive Cancer Network®. 2023; Accessed May 2024:
- Else T, Greenberg S, Fishbein L. Hereditary Paraganglioma-Pheochromocytoma Syndromes. [Updated 2023 Sep 21]. GeneReviews® [Internet]. 2023.
- NCCN. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Kidney Cancer Version 4.2023. National Comprehensive Cancer Network®. 2023; Accessed May 2024.
- MacMahon H, Naidich D P, Goo J M, Lee K S, Leung A N C et al. Guidelines for Management of Incidental Pulmonary Nodules Detected on CT Images: From the Fleischner Society 2017. Radiology. 2017; 284: 228-243. 10.1148/radiol.2017161659.
Coding Section
| Code | Number | Description |
| CPT | 71250 | Computed tomography, thorax; without contrast material |
| 71260 | Computed tomography, thorax; with contrast material(s) | |
| 71270 | Computed tomography, thorax; without contrast material, followed by contrast material(s) and further sections | |
| 71271 | Computed tomography, thorax; low dose for lung cancer screening; without contrast material(s) |
Procedure and diagnosis codes on Medical Policy documents are included only as a general reference tool for each policy. They may not be all-inclusive.
This medical policy was developed through consideration of peer-reviewed medical literature generally recognized by the relevant medical community, U.S. FDA approval status, nationally accepted standards of medical practice and accepted standards of medical practice in this community, and other nonaffiliated technology evaluation centers, reference to federal regulations, other plan medical policies, and accredited national guidelines.
"Current Procedural Terminology © American Medical Association. All Rights Reserved"
History From 2019 Forward
| 12/02/2024 | Annual review, policy reformatted for clarity and consistency updating: Lung Cancer screening is consistent with Cancer society • Lung nodules sections was clarified for size and follow up studies • Infections and inflammation section added to incorporate indications within the GL that were alone and added in sarcoidosis • Reorganized the malignancy section to follow the abdomen GLs; for known malignancy Initial staging was broad, Restaging gave the situations not reasonable, and surveillance was each identified with timelines for acceptable studies • Genetic Syndromes and Rare Diseases was added/adjusted. • Combination Studies were expanded upon to coincide with other guidelines/combination studies. Also adding purpose, contraindications/preferred studies, rationale and references. |
| 11/30/2023 | Annual review, entire policy updated for consistency. Updated Covid information, clarified details on nudules seen in other imaging. Added tranplant imaging. |
| 12/16/2022 | Annual review, addition of single ventricle heart disease coverage criteria, No other changes. |
| 12/15/2021 |
Annual review updating existing information regarding Fleischner criteria and Lung Rads. Also adding section related to COVID 19. Updating description and references. |
| 12/10/2020 |
Updated coding with 2021 codes. No other changes. |
| 12/01/2020 |
Annual review, policy updated for clarity and also adding verbiage regarding low dose CT scanning previously in CAM 60130. Entire policy updated to encompass that addition. |
| 12/03/2019 |
New Policy |