Folate Testing - CAM 200
Description
Folate, or vitamin B9, is a generic term for a water-soluble vitamin obtained from the diet that is involved in the transfer of methyl groups (i.e., single carbon-containing groups) in multiple biochemical metabolic pathways, including nucleic acid biosynthesis and methionine/homocysteine metabolism. Folate metabolism is closely linked to vitamin B12, cobalamin. Folate deficiency can be implicated in many disease states and processes; however, it is usually easily remedied with either a change in diet or a dietary supplement of the synthetic form, folic acid.1,2
Regulatory Status
The FDA has approved multiple tests for the evaluation of both human serum folate and red blood cell folate levels either alone, together, or included in panels. As of March 27, 2020, there are 83 records on the FDA Devices database for folate.
Additionally, many labs have developed specific tests that they must validate and perform in house. These laboratory-developed tests (LDTs) are regulated by the Centers for Medicare & Medicaid Services (CMS) as high-complexity tests under the Clinical Laboratory Improvement Amendments of 1988 (CLIA ’88). As an LDT, the U.S. Food and Drug Administration has not approved or cleared this test; however, FDA clearance or approval is not currently required for clinical use.
Policy
Application of coverage criteria is dependent upon an individual’s benefit coverage at the time of the request.
- Measurement of serum folate concentration is considered MEDICALLY NECESSARY in any of the following situations:
- For individuals with megaloblastic or macrocytic anemia.
- For individuals who have undergone, or for those who have been scheduled for, bariatric procedures such as Roux-en-Y gastric bypass, sleeve gastrectomy, or biliopancreatic diversion/duodenal switch.
- For all indications not described above, measurement of serum folate concentration is considered NOT MEDICALLY NECESSARY.
- For all indications, measurement of red blood cell (RBC) folate is considered NOT MEDICALLY NECESSARY.
The following does not meet coverage criteria due to a lack of available published scientific literature confirming that the test(s) is/are required and beneficial for the diagnosis and treatment of an individual’s illness.
- For all situations, folate receptor autoantibody testing is considered NOT MEDICALLY NECESSARY.
Table of Terminology
| Term |
Definition |
| AA |
Aplastic anemia |
| AACE |
American Association of Clinical Endocrinologists |
| AAFP |
American Academy of Family Physicians |
| AAN |
American Academy of Neurology |
| ABIM |
American Board of Internal Medicine |
| ACE |
American College of Endocrinology |
| ACG |
American College of Gastroenterology |
| AND |
Academy of Nutrition and Dietetics |
| ApABG |
Para-acetamidobenzoylglutamate |
| ASA |
American Society of Anesthesiologists |
| ASCP |
American Society for Clinical Pathology |
| ASMBS |
American Society for Metabolic and Bariatric Surgery |
| ASPEN |
American Society for Parenteral and Enteral Nutrition |
| BCMA |
British Columbia Medical Association |
| BCSH |
British Committee for Standards in Haematology |
| CBC |
Complete blood count |
| CD |
Celiac disease |
| CDC |
Centers for Disease Control and Prevention |
| CKD |
Chronic kidney disease |
| CLIA ’88 |
Clinical Laboratory Improvement Amendments of 1988 |
| CMS |
Centers for Medicare & Medicaid Services |
| CPT |
Current Procedural Terminology |
| CRP |
C-reactive protein |
| DFE | Dietary folate equivalents |
| DHFR |
Dihydrofolate reductase |
| DNA |
Deoxyribonucleic acid |
| ECCO |
European Crohn’s and Colitis Organization |
| EL |
Evidence level |
| FDA |
Food and Drug Administration |
| Hb |
Hemoglobin |
| HCPCS |
Healthcare Common Procedure Coding System |
| IBD |
Irritable bowel disorders |
| IOM |
Institute of Medicine |
| KDIGO |
Kidney Disease Improving Global Outcomes |
| LDT |
Laboratory-developed test |
| MCG |
Microgram |
| MCMs |
Major congenital malformation |
| MMWR |
Morbidity and mortality weekly report |
| MTHFR |
Methylenetetrahydrofolate reductase |
| NBA |
Australian National Blood Authority |
| NCCN |
National Comprehensive Cancer Network |
| NHANES |
National Health and Nutrition Examination Survey |
| NICE |
National Institute for Health and Care Excellence |
| NTDs |
Neural tube defects |
| OMA |
Obesity Medicine Association |
| PA |
Prior authorization |
| pABG |
Para-aminobenzoylglutamate |
| RBC |
Red blood cell |
| RDN |
Registered dietitian nutritionist |
| RF |
Red blood cell folate testing |
| SCCM |
Society of Critical Care Medicine |
| SF |
Serum folate testing |
| SSCM |
Society of Critical Care Medicine |
| TOS |
The Obesity Society |
| TSAT |
Transferrin saturation |
| UL |
Upper limits |
| USPSTF |
United States Preventive Services Task Force |
Rationale
Folate, or vitamin B9, naturally occurs as polyglutamated compounds (pteroylpolyglutamates) in many plant and animal products. The synthetic form is a monoglutamate-containing compound called folic acid. Folic acid is more chemically stable for commercial production and storage, but it is less bioavailable than the naturally occurring folate.2 Biochemically, folate is a coenzyme in single carbon transfers in vivo and is directly linked to the cobalamin (vitamin B12) cycle, methionine metabolism, and nucleic acid biosynthesis. Dietary folates are hydrolyzed via γ-glutamyl hydrolase (or folate conjugase) prior to absorption in the intestinal mucosa.3 Both folate and vitamin B12 are required for formation of 5,10-methylene tetrahydrofolate, which is the cofactor involved in purine synthesis.
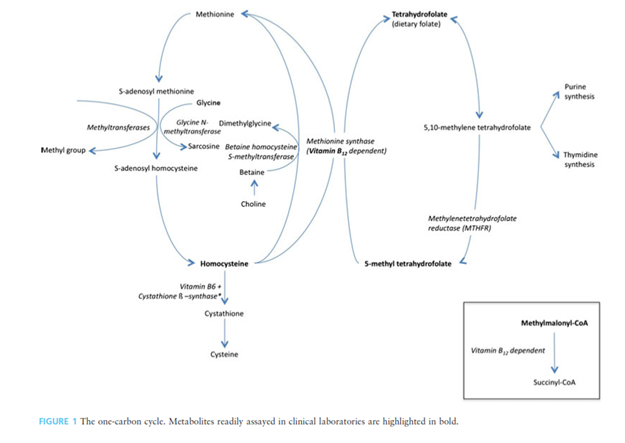
Methylenetetrahydrofolate reductase (MTHFR) is the enzyme responsible in converting 5,10-methylene tetrahydrofolate to 5-methyl tetrahydrofolate, which is required for methionine synthase, the enzyme that converts homocysteine to methionine. The interlinked one-carbon cycle is depicted in the figure below with the metabolites assayed in clinical laboratories in bold.4
Role of Folate in Anemia
Anemia occurs when the body lacks healthy red blood cells (RBCs), leading to an insufficient amount of oxygen delivered to tissues. Typical symptoms of anemia include fatigue, weakness, pale skin, and lightheadedness.
Macrocytic anemia refers to anemias that have high mean corpuscular volume with large RBCs. Mean corpuscular volume, or mean cell volume, can be defined as the average volume of RBCs in an individual. Megaloblastic anemia is a specific macrocytic anemia due to nucleic acid metabolic defects that result in “nuclear-cytoplasmic dyssynchrony, reduced number of cell divisions in the bone marrow, and nuclear abnormalities in both myeloid and erythroid precursors” caused by folate and/or vitamin B12 deficiency.5 These abnormal RBCs are the principle clinical manifestations of folate deficiency and symptoms “include weakness, fatigue, difficulty concentrating, irritability, headache, heart palpitations, and shortness of breath.”1
Folate and Neural Tube Defects (NTDs)
Neural tube defects (NTDs) develop early in pregnancy and are malformations of the brain and/or spine that include spina bifida and anencephaly. Folate deficiency is directly linked to NTDs. The role of folate in NTD development is not well-characterized. The role of folate in either the methylation cycle or nucleic acid synthesis has been suggested to play a part in NTD development during embryogenesis, and some studies have indicated that it is the bioavailability of specific folates in the pregnant individual that can increase the likelihood of NTDs.6,7 Individuals typically do not obtain enough folate from diet alone, so individuals of childbearing age are recommended to take a synthetic folic acid supplement to decrease the likelihood of NTDs in offspring.8 To decrease the occurrence of NTDs and folate deficiency, the United States and Canada mandated folic acid supplementation to cereal grains in 1998, and as of March 2018 “94 countries have legislation to mandate fortification of at least one industrially milled cereal grain.”9 Nutrition labels in the U.S. now display folate as mcg dietary folate equivalents (mcg DFE) alongside folic acid amount to clarify dietary intake.10
It is notable that the prevalence of folate deficiency, and the prevalence of NTDs has declined in countries with routine folic acid supplementation.11 A review by Imbard, et al. (2013) of 17 different studies on the impact of folic acid fortification of NTD rates show that 16 show a decrease in the rate of NTDs. Only one study of the rate of NTDs in California showed no decline since fortification. The reduction of the United States overall was 26-30% since folic acid fortification.6
Folate Receptor Antibody Testing (FRAT®)
Folate deficiency in the pregnant individual can “lead to pregnancy-related complications including NTDs in the fetus. Numerous studies have now established the benefits of folate supplementation in reducing the incidence of NTD pregnancy.”12 Fratnow's FRAT® is a diagnostic blood test that screens for autoantibodies to the Folate Receptor Alpha, known as folate receptor autoantibodies. The presence of folate receptor autoantibodies suggests that folate (vitamin B9), which is an essential vitamin for proper growth and neurological function, is not being properly transported to the brain and into cerebrospinal fluid (CSF), among other tissues where Folate Receptor Alpha may be found. “The early detection of folate receptor autoantibodies is necessary because it can lead to timely diagnosis and appropriate management of conditions associated with these autoantibodies. Folate receptor autoantibodies have been found in Cerebral Folate Deficiency Syndrome, Autism Spectrum Disorders, Schizophrenia, Depression and Neural Tube Defects, among other neurodevelopmental disorders.”13
Causes of Folate Deficiency
Folate deficiency can be caused by dietary intake. Nutritional deficits may occur due to diet, alcoholism, depression, and even overcooked foods. Many malabsorptive disorders, such as celiac disease and ulcerative colitis, can also result in a decrease in folate uptake. Further, bariatric procedures may result in decreased absorption, and drugs, including methotrexate and trimethoprim that inhibit dihydrofolate reductase (DHFR), can also cause a folate deficiency. It is also important to note that an increased need of folate for DNA synthesis during pregnancy and lactation, chronic hemolytic anemias, exfoliative skin diseases, and hemodialysis cause folic acid deficiency. Folate deficiency is also more prevalent in older adults than younger.5
Methodology of Folate Testing
Folate concentrations have been measured from serum, erythrocytes (RBC), and urine. Serum folate levels may not “differentiate between what may be a transitory reduction in folate intake or chronic folate deficiency accompanied by depleted folate stores and functional changes.”3 RBCs have a lifespan of approximately 120 days, and folate is only taken in during initial erythropoiesis (red blood cell production); consequently, RBC folate concentrations are less likely to be affected by transitory dietary fluctuations. However, Wu, et al. (1975) show that both RBC folate and serum folate levels correlate to hepatocyte folate levels.3,14 Galloway and Rushworth (2003) released a study in conjunction with the National Pathology Alliance review in the United Kingdom comparing data of laboratories of the National Health Service that routinely use serum folate testing only, RBC folate testing only, or both serum and RBC folate testing together. The researchers conclude that there is no need to use both tests to determine folate concentration as an initial screen. “The serum folate assay provided equivalent information to the measurement of red cell folate and evidence from the literatures [sic] suggest that the serum folate assay should be the method of choice.”15
Clinical Utility and Validity
A study by Shojania and von Kuster (2010) investigated the use of serum folate testing (SF) and RBC folate testing (RF) in cases of anemia in a country that has mandated folic acid supplementation in grain products. By examining the data for folate testing in anemia at two different teaching hospitals in Canada, they report that in one hospital in 2001 “11 out of 2154 (0.5%) SF were low (<7.0 nmol/L) and four out of 560 (0.7%) RF were low (<417 nmol/L). In no subject with low SF or RF could the anemia be attributed to folate deficiency.” For the other hospital, the data from 1999-2001 shows that “19 out of 991 (1.9%) had low RF (<225 nmol/L) but in only 2 patients (0.2%) the low RF was in folate deficiency anemia range.”16 The authors conclude that neither serum folate testing, nor RBC folate testing is justified in cases of anemia for folic acid fortified countries due to such low incidence rates of folate deficiency anemia.
A study by Joelson, et al. (2007) examined the records of three different hospitals in the U.S. that service a high number of indigent patients. The researchers reported the data from three non-consecutive years (1997, 2000, and 2004) to examine the impact of folate fortification in food products. Using the RBC folate levels only with a RBC folate cutoff value of 160 ng/mL (363.6 nmol/L), “the combined incidence of folate deficiency decreased from 4.8% in 1997 to 0.6% in 2004…Even when the folate concentration was found to be low, the majority of these subjects did not have macrocytosis.” This study included a total of 4134 RBC folate tests performed over the course of three years. It is of interest to note that the number of tests performed increased from 813 in 1997 to 1759 in 2004. The authors do note of a potential limitation of the study since the data of the patients cannot be separated into specific groups (pregnant individuals, alcoholics, socioeconomic classes, and so on). The authors conclude “that folate deficiency has become a rare event in the United States, and the utility of routine folate measurements for patients with anemia and/or increased mean corpuscular volume are difficult to justify.”17
Urinary folate levels do not reflect either the stored folate concentrations or the fluctuations in folate concentration due to transitory dietary changes. Only about one to two percent of the folate excreted in the urine is unmetabolized and “excretion continued in the face of advanced folate depletion.”3 One study of ten postmenopausal individuals on a low folate diet measured folate turnover using urinary testing of folate and folate metabolites. “Folate intake did not significantly influence ApABG (para-acetamidobenzoylglutamate) or pABG (para-aminobenzoylglutamate) excretion.” ApABG and pABG along with pterins are the major folate catabolites. The authors conclude that “the rate of folate catabolite excretion is related mainly to masses of slow-turnover folate pools governed by long-term folate intake.”18
Epstein-Peterson, et al. (2020) collected and analyzed all folate tests performed in 2017 at an academic cancer center. In total, 937 patients were tested 1065 times; approximately seven percent of tests indicated a folate deficiency, and folate deficiency was significantly associated with a higher risk of death (P=0.01).19
Tran, et al. (2022) performed a literature review on the diagnostic accuracy, clinical utility, cost-effectiveness, and evidence-based guidelines regarding the use of serum folate testing in people with suspected folate deficiency. An information specialist completed a literature search using the search concepts “folate deficiency AND testing” and only limiting results to the human population for publications between January 1, 2012, and February 15, 2022. The authors were not able to identify any relevant literature regarding diagnostic test accuracy, clinical utility, cost-effectiveness, or evidence-based guidelines.20
Centers for Disease Control and Prevention
The CDC urges all individuals who are capable of becoming pregnant and who are also of reproductive age to “ get 400 mcg of folic acid every day. . . Getting 400 micrograms of folic acid daily can help prevent a type of serious birth defects called neural tube defects.”21 This recommendation includes all individuals of reproductive age planning to become pregnant or not, as about half of U.S. pregnancies are unplanned. They also recommend that individuals who have had a previous pregnancy affected by NTD and plan to become pregnant again take 4,000 mcg of folic acid daily. Folic acid should be started at least one month before conception and continue through the first three months of pregnancy.
For individuals with a history of an NTD-affected pregnancy who are not planning to become pregnant, it is recommended that they take 400 mcg of folic acid daily.10
American Society for Clinical Pathology/Choosing Wisely
The ASCP published a recommendation in 2017 in Choosing Wisely, an American Board of Internal Medicine (ABIM) initiative, where they clearly state the following: “Do not order red blood cell folate levels at all. In adults, consider folate supplementation instead of serum folate testing in patients with macrocytic anemia.” They indicate that the drastic decrease in folic deficiency in both the U.S. and Canada after mandated folic acid supplementation in foods no longer requires for either serum folate or red blood cell folate concentrations be tested. “While red blood cell folate levels have been used in the past as a surrogate for tissue folate levels or a marker for folate status over the lifetime of red blood cells, the result of this testing does not, in general, add to the clinical diagnosis or therapeutic plan.”22
National Pathology Alliance (of the United Kingdom)
The National Pathology Alliance of the United Kingdom in 2003 published in the Journal of Clinical Pathology their recommendation “that serum folate measurements provide equivalent information to red cell folate measurements.”15
American Association of Clinical Endocrinologists (AACE)/The American College of Endocrinology (ACE), The Obesity Society (TOS), American Society for Metabolic and Bariatric Surgery (ASMBS), Obesity Medicine Association (OMA), and American Society of Anesthesiologists (ASA)
In 2013, the AACE, ACE, and TOS issued joint guidelines regarding healthy eating for the prevention and treatment of metabolic and endocrine diseases in adults.23 Based on the data from the National Health and Nutrition Examination Survey (NHANES), they state “that patients with vitamin B12 deficiency had higher folate levels, were more likely to be anemic, and had more cognitive impairment that those with normal serum folate levels” [evidence level (EL) 2]. They evaluate the evidence concerning the link between folate and cardiovascular disease as EL4 and the link between NTDs and folate as EL1. With respect to pregnancy nutritional needs, they “should be assessed prior to conception to improve pregnancy outcome.” All individuals of childbearing age “should consume at least 400 µg dietary equivalents of folate per day” [EL4] and that during pregnancy the daily amount should be increased to 600 µg [EL3].
The AACE and ACE in 2015 released their Clinical Practice Guidelines for Developing a Diabetes Mellitus Comprehensive Care Plan.24 Concerning patients with diabetic nephropathy, the suggest that they “undergo annual or more frequent assessment of electrolytes.” For those with anemia, iron, transferrin saturation (TSAT), ferritin, vitamin B12, and folate levels “should be further investigated” [EL4]. In the 2022 AACE update, the guidelines suggest that persons with chronic kidney disease in diabetes mellitus should undergo annual or more frequent assessment of blood counts to assess anemia status as this should be further investigated with iron, TSAT, ferritin, vitamin B12, and folate levels.25
In 2017, the AACE and ACE released their guidelines for management of dyslipidemia and prevention of cardiovascular disease.26 Since bile acid sequestrant treatments such as cholestyramine can cause folate depletion in children, they recommend that children on such treatments supplement their diet with a multivitamin. They also note that folate, B6, and B12 supplementation can help mediate hyperhomocysteinemia, but that the supplements do not reduce risk of atherosclerotic cardiovascular disease.
In 2019, the AACE/ACE, TOS, ASMBS, OMA, and ASA issued joint guidelines for the perioperative nutritional, metabolic, and nonsurgical support of the bariatric surgery patient.27 Here, as part of a pre-operative bariatric surgery checklist that has a “Grade A” recommendation, they include “nutrient screening with iron studies, B12, and folic acid (RBC folate, homocysteine, methylmalonic acid optional) …consider more extensive testing in patients undergoing malabsorptive procedures based on symptoms and risks.” With regards to patients who become pregnant after having a bariatric procedure, they recommend (with Grade D) having nutritional surveillance laboratory screenings done each trimester for folate deficiency along with iron, calcium, B12, and vitamin D, and if after a malabsorptive procedure, fat-soluble vitamins, zinc, and copper. With a Grade C, they state that “nutritional anemias resulting from malabsorptive bariatric surgical procedures can involve deficiencies in vitamin B12, folate, protein, copper, selenium, and zinc and may be evaluated when routine aggressive case finding for iron-deficiency anemia is negative.” Additionally, findings of folate deficiency in patients with obesity prior to bariatric surgery by the ASMBS “justifies aggressive case finding preoperatively with biochemical testing, specifically using sensitive markers, such as red-blood-cell folate and homocysteine (methylmalonic acid is normal with folate deficiency and normal B12 status)” and they note that particular attention should be given to individuals of childbearing age.
National Institute for Health and Care Excellence
The National Institute for Health and Care Excellence (NICE) of the Department of Health in the United Kingdom published their extensive guidelines concerning bladder cancer on February 25, 2015. Within the section concerning the follow-up treatment for muscle-invasive bladder cancer, they recommend a protocol after radical cystectomy that includes “monitoring for metabolic acidosis and B12 and folate deficiency at least annually.”28 This guideline was reaffirmed in 2019.
In a guideline on maternal and child nutrition, NICE recommends that the importance of folic acid supplementation should be discussed with anyone who is or may become pregnant. They recommend that healthcare workers “Advise anyone who may become, or is planning to become, pregnant or is in the first 12 weeks of pregnancy to take 400 micrograms of folic acid a day.” Additionally, it is recommended to “Offer a high-dose folic acid supplement (5 mg a day) to anyone who is planning to become pregnant or is in the first 12 weeks of pregnancy if they have an increased risk of having a baby with a neural tube defect or other congenital malformation, for example, if they:
- (or their partner) have, or if there is a family history of, a neural tube defect or other congenital malformation.
- have had a previous pregnancy affected by a neural tube defect or other congenital malformation.
- have type 1 or type 2 diabetes.
- have a haematological condition that requires folic acid supplementation, such as sickle cell anaemia or thalassaemia.
- are taking medicines that can affect how folic acid is absorbed or metabolised (for example, people taking anti-epileptic medicines or medicines for HIV).”29
American Academy of Family Physicians (AAFP)
Although the American Academy of Family Physicians (AAFP) published a 2009 diagnostic algorithm for macrocytic anemia that favored red blood cell folate testing over serum folate,30 this recommendation is no longer followed and has been superseded by more recent guidance. More recent AAFP-endorsed recommendations include participating in the Choosing Wisely campaign which advises against ordering red blood cell folate testing. The recommendation is to consider folate supplementation instead of serum folate testing in patients with macrocytic anemia.31
American Academy of Neurology
In 2001, the AAN updated their practice parameters for the diagnosis of dementia. Within the section concerning the comorbidities that should be screened in an initial assessment for dementia, they recommend folate testing along with complete blood count, serum electrolytes, B12, blood urea nitrogen/creatinine, syphilis serology, thyroid function, and glucose. They did note that as of that time “no studies were identified that evaluated these recommendations” since the last practice parameters released in 1994.32
Kidney Disease Improving Global Outcomes (KDIGO)
The KDIGO released their updated KDIGO Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease in 2026. They note that folate deficiency has been linked as a cause for anemia in CKD. They gave a “not graded” recommendation for “In people with anemia and CKD in whom the initial tests do not reveal the cause, consider an expanded panel to identify potential underlying causes as warranted based on the clinical scenario:
- Blood smear review
- Haptoglobin
- Lactate dehydrogenase
- C-reactive protein
- Vitamin B12
- Folate
- Liver function tests
- Serum protein electrophoresis with immunofixation, serum free light chains, and urinary Bence-Jones protein
- thyroid-stimulating hormone
- Parathyroid hormone
- Fecal occult blood test.“33
American Society for Parenteral and Enteral Nutrition (ASPEN) & Society of Critical Care Medicine (SSCM)
In 2013, ASPEN and SSCM issued joint clinical guidelines concerning the nutrition support of hospitalized obese adults. With a “Recommendation: Weak” status, they recommended “in acutely ill hospitalized patients with history of these procedures [sleeve gastrectomy, gastric bypass, or biliopancreatic diversion ± duodenal switch], evaluation for evidence of depletion of iron, copper, zinc, selenium, thiamine, folate, and vitamins B12 and D is suggested as well as repletion of deficiency states.”34
In 2016, ASPEN and SSCM issued their guidelines for the provision and assessment of nutrition support therapy in the adult critically ill patient. The committee recommended that “evaluation for and treatment of micronutrient deficiencies such as calcium, thiamin, vitamin B12, fat-soluble vitamins (A, D, E, K), and folate, along with the trace minerals iron, selenium, zinc, and copper, should be considered.”35 In 2021, the ASPEN and SSCM updated their guidelines for the provision and assessment of nutrition support therapy in the adult critically ill patient; the guidelines do not mention folate testing.36 In 2017, ASPEN and SSCM updated their guidelines for the provision and assessment of nutrition support therapy in the pediatric critically ill patient. These guidelines do not mention folate testing.37
Academy of Nutrition and Dietetics (AND)
The Academy of Nutrition and Dietetics released their oncology evidence-based nutrition practice guideline in 2013 and reaffirmed the guideline in a 2017 publication. On the “Assessment of Biochemical Data Medical Tests, and Procedures on Adult Oncology Patients” portion, the committee recommended with “Consensus, Imperative” that “the RDN [Registered Dietitian Nutritionist] should evaluate available data and recommend as indicated: biochemical data, medical tests and procedures of adult oncologypatients” and included on their list is “Nutritional anemia profile (hemoglobin, hematocrit, folate, B12, iron).” “Assessment of these factors is needed to effectively determine nutrition diagnoses and plan the nutrition interventions.”38
European Crohn’s and Colitis Organisation (ECCO)
The ECCO’s guidelines concerning irritable bowel disorders (IBD) included an extensive discussion on causes and treatments of anemia in IBD—both iron deficiency anemia and non-iron deficiency anemia. With an [EL 5], they state that “deficiencies of Vitamin B12 and folate should be treated to avoid anaemia. Serum levels of vitamin B12 and folic acid should be measured at least annually, or if macrocytosis is present. Patients at risk for vitamin B12 or folic acid deficiency [eg small bowel disease or resection] need closer surveillance. The recommended timelines are based on expert opinions and reflect common clinical practice, but do not apply to patients with extensive small bowel resection, extensive ileal Crohn’s disease, or ileal-anal pouch.”39
American College of Gastroenterology (ACG)
In their guidelines and recommendations concerning the diagnosis and management of celiac disease (CD) in 2013, the ACG recommended the following statement with Conditional recommendation, low level of evidence: “People with newly diagnosed CD should undergo testing and treatment for micronutrient deficiencies. Deficiencies to be considered for testing should include, but not be limited to, iron, folic acid, vitamin D, and vitamin B12.”40 This guideline was reaffirmed in 2016.
The ACG updated its guidelines for the diagnosis and management of celiac disease in 2023 and stated that an approach to monitoring celiac disease includes “tissue transglutaminase and deamidated gliadin peptide can be used for monitoring celiac disease considering the availability of test at baseline before initiation of the gluten-free diet. Other tests may include complete blood count, alanine aminotransferase, aspartate aminotransferase, vitamins (A, D, E, B12), copper, zinc, folic acid, ferritin, and iron.” The ACG goes on to say that blood tests at follow-up appointments should be individualized to verify correction of lab tests that were abnormal at baseline.41
British Committee for Standards in Haematology
In 2014, the BCSH released guidelines on folate deficiencies. They noted that “routine red cell folate testing is not necessary because serum folate alone is sufficient in most cases.” However, they also acknowledged that “in the presence of strong clinical suspicion of folate deficiency, despite a normal serum level, a red cell folate assay may be undertaken, having ruled out cobalamin deficiency.” The BCSH also noted that “folate status is generally checked in clinical situations similar to those of cobalamin deficiency (Grade 1A).”42
In 2016, the BCSH recommended that a “documented vitamin B12 or folate deficiency should be corrected before a final diagnosis of AA is confirmed. Bone marrow aplasia due to vitamin deficiency is exceedingly rare.”43
In the 2021 BCSH Guidelines for the Investigation and Management of Vitamin B12 and Folate Deficiency list the following four indications for folate testing: “unexplained anaemia/macrocytic anaemia/megaloblastic anaemia, excess alcohol intake especially with coexisting liver disease, exfoliative skin diseases, post gastric and bariatric surgery.” Alternatively, the guidelines list the following two indications when folate supplementation should occur without folate testing: “pregnancy, haemolytic anaemia haemoglobinopathies.” The guidelines also state that folate and B12 should always be tested together, but notes that “once a patient has commenced B12 replacement there is no further need for it to be measured again.”44
Renal Association Clinical Practice Guideline
The Renal Association recommends measuring serum folate concentration for evaluation of anemia in CKD.45
National Comprehensive Cancer Network
The NCCN recommends measurement of serum or RBC folate as part of the initial evaluation for cytopenia(s) that are suspected to be myelodysplastic syndromes. NCCN also notes that iron, folate, or vitamin B12 levels should be checked as needed to rule out coexisting causes of anemia in the management of myelodysplastic syndrome-related anemia.46
References
- NIH. Folate Dietary Supplement Fact Sheet. National Institutes of Health. Updated November 30, 2022. https://ods.od.nih.gov/factsheets/Folate-HealthProfessional/
- Means Jr RT, Fairfield KM. Causes and pathophysiology of vitamin B12 and folate deficiencies. Updated october 2, 2025. https://www.uptodate.com/contents/causes-and-pathophysiology-of-vitamin-b12-and-folate-deficiencies
- IOM. The National Academies Collection: Reports funded by National Institutes of Health. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. National Academies Press (US) National Academy of Sciences.; 1998.
- Finer S, Saravanan P, Hitman G, Yajnik C. The role of the one‐carbon cycle in the developmental origins of Type 2 diabetes and obesity. Diabetic Medicine. March 01, 2014 2013;31(3):263-272. doi:10.1111/dme.12390
- Means Jr RT, Fairfield KM. Clinical manifestations and diagnosis of vitamin B12 and folate deficiency. Updated December 16, 2025. https://www.uptodate.com/contents/clinical-manifestations-and-diagnosis-of-vitamin-b12-and-folate-deficiency
- Imbard A, Benoist J-F, Blom HJ. Neural Tube Defects, Folic Acid and Methylation. International Journal of Environmental Research and Public Health. 2013;10(9):
4352-4389 - Rothenberg SP, da Costa MP, Sequeira JM, et al. Autoantibodies against Folate Receptors in Women with a Pregnancy Complicated by a Neural-Tube Defect. New England Journal of Medicine. January 08, 2004 2004;350(2):134-142. doi:10.1056/NEJMoa031145
- Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Folic Acid Supplementation for the Prevention of Neural Tube Defects: US Preventive Services Task Force Recommendation Statement. Jama. 2017;317(2):183-189. doi:10.1001/jama.2016.19438
- Food Fortification Initiative. Global Progress. https://ffinetwork.org/global-progress/
- CDC. Folic Acid: Facts for Clinicians. Updated May 20, 2025. https://www.cdc.gov/folic-acid/hcp/clinical-overview/index.html
- Crider KS, Bailey LB, Berry RJ. Folic acid food fortification-its history, effect, concerns, and future directions. Nutrients. 2011;3(3):370-84. doi:10.3390/nu3030370
- Sequeira JR, Vincent Quadros, Edward. The diagnostic utility of folate receptor autoantibodies in blood 2012;doi:10.1515/cclm-2012-0577
- Fratnow. What is FRAT? https://www.fratnow.com/
- Wu A, Chanarin I, Slavin G, Levi AJ. Folate Deficiency in the Alcoholic—its Relationship to Clinical and Haematological Abnormalities, Liver Disease and Folate Stores. British Journal of Haematology. March 01, 1975 1975;29(3):469-478. doi:10.1111/j.1365-2141.1975.tb01844.x
- Galloway M, Rushworth L. Red cell or serum folate? Results from the National Pathology Alliance benchmarking review. Journal of clinical pathology. 2003;56(12):924-6. doi:10.1136/jcp.56.12.924
- Shojania AM, von Kuster K. Ordering folate assays is no longer justified for investigation of anemias, in folic acid fortified countries. BMC Research Notes. 2010;3:22-22. doi:10.1186/1756-0500-3-22
- Joelson DW, Fiebig EW, Wu AH. Diminished need for folate measurements among indigent populations in the post folic acid supplementation era. Archives of pathology & laboratory medicine. Mar 2007;131(3):477-80. doi:10.5858/2007-131-477-DNFFMA
- Gregory IIIJF, Swendseid ME, Jacob RA. Urinary Excretion of Folate Catabolites Responds to Changes in Folate Intake More Slowly than Plasma Folate and Homocysteine Concentrations and Lymphocyte DNA Methylation in Postmenopausal Women. The Journal of Nutrition. 2000;130(12):2949-2952. doi:10.1093/jn/130.12.2949
- Epstein-Peterson ZD, Li DG, Lavery JA, Barrow B, Chokshi I, Korenstein D. Inpatient folate testing at an academic cancer center: single-year experience. Support Care Cancer. January 03, 2020 2020;doi:10.1007/s00520-019-05267-1
- Tran K, Mierzwinski-Urban M, Mahood Q. Folate Testing in People With Suspected Folate Deficiency. Canadian Journal of Health Technologies. 2022;2(3)doi:10.51731/cjht.2022.295
- CDC. Folic Acid. Updated May 20, 2025 https://www.cdc.gov/folic-acid/about/
- ASCP. ASCP News. Critical Values. 2017;11(1):34-39. doi:10.1093/crival/vax040
- Gonzalez-Campoy JM, St Jeor ST, Castorino K, et al. Clinical practice guidelines for healthy eating for the prevention and treatment of metabolic and endocrine diseases in adults: cosponsored by the American Association of Clinical Endocrinologists/the American College of Endocrinology and the Obesity Society. Endocrine practice : official journal of the American College of Endocrinology and the American Association of Clinical Endocrinologists. 2013;19 Suppl 3:1-82. doi:10.4158/ep13155.gl
- Handelsman Y, Bloomgarden ZT, Grunberger G, et al. American association of clinical endocrinologists and american college of endocrinology - clinical practice guidelines for developing a diabetes mellitus comprehensive care plan - 2015. Endocrine practice : official journal of the American College of Endocrinology and the American Association of Clinical Endocrinologists. 2015;21 Suppl 1:1-87. doi:10.4158/ep15672.Gl
- Blonde L, Umpierrez GE, Reddy SS, et al. American Association of Clinical Endocrinology Clinical Practice Guideline: Developing a Diabetes Mellitus Comprehensive Care Plan-2022 Update. Endocrine practice : official journal of the American College of Endocrinology and the American Association of Clinical Endocrinologists. 2022;28(10):923-1049. doi:10.1016/j.eprac.2022.08.002
- Jellinger PS, Handelsman Y, Rosenblit PD, et al. American Association of Clinical Endocrinologists and American College of Endocrinology Guidelines for Management of Dyslipidemia and Prevention of Cardiovascular Disease. Endocrine practice : official journal of the American College of Endocrinology and the American Association of Clinical Endocrinologists. Apr 2017;23(Suppl 2):1-87. doi:10.4158/ep171764.appgl
- Mechanick JI, Apovian C, Brethauer S, et al. Clinical Practice Guidelines For The Perioperative Nutrition, Metabolic, And Nonsurgical Support Of Patients Undergoing Bariatric Procedures - 2019 Update: Cosponsored By American Association Of Clinical Endocrinologists/American College Of Endocrinology, The Obesity Society, American Society For Metabolic & Bariatric Surgery, Obesity Medicine Association, And American Society Of Anesthesiologists - Executive Summary. Endocrine practice : official journal of the American College of Endocrinology and the American Association of Clinical Endocrinologists. 2019;25(12):1346-1359. doi:10.4158/gl-2019-0406
- Bladder cancer: diagnosis and management (National Institute for Health and Care Excellence) (2015).
- NICE. Maternal and child nutrition: nutrition and weight management in pregnancy, and nutrition in children up to 5 years. Updated January 15, 2025. https://www.nice.org.uk/guidance/ng247/chapter/Recommendations
- Kaferle J, Strzoda CE. Evaluation of macrocytosis. American family physician. 2009;79(3):203-8.
- AAFP. Choosing Wisely Recommendations. American Academy of Family Physicians. https://www.aafp.org/pubs/afp/collections/choosing-wisely/350.html
- Knopman DS, DeKosky ST, Cummings JL, et al. Practice parameter: diagnosis of dementia (an evidence-based review). Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology. 2001;56(9):1143-53. doi:10.1212/WNL.56.9.1143
- KDIGO. KDIGO 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease (CKD). Kidney International. 2026;109(1):S1-S99. doi:10.1016/j.kint.2025.06.006
- Choban P, Dickerson R, Malone A, Worthington P, Compher C. A.S.P.E.N. Clinical Guidelines: nutrition support of hospitalized adult patients with obesity. Journal of Parenteral and Enteral Nutrition. Novemeber 01, 2013 2013;37(6):714-744. doi:10.1177/0148607113499374
- McClave SA, Taylor BE, Martindale RG, et al. Guidelines for the Provision and Assessment of Nutrition Support Therapy in the Adult Critically Ill Patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.). JPEN J Parenter Enteral Nutr. 2016;40(2):159-211. doi:10.1177/0148607115621863
- Compher C, Bingham AL, McCall M, et al. Guidelines for the provision of nutrition support therapy in the adult critically ill patient: The American Society for Parenteral and Enteral Nutrition. JPEN J Parenter Enteral Nutr. 2022;46(1):12-41. doi:10.1002/jpen.2267
- Mehta NM, Skillman HE, Irving SY, et al. Guidelines for the Provision and Assessment of Nutrition Support Therapy in the Pediatric Critically Ill Patient: Society of Critical Care Medicine and American Society for Parenteral and Enteral Nutrition. Pediatr Crit Care Med. 2017;18(7):675-715. doi:10.1097/pcc.0000000000001134
- Thompson KL, Elliott L, Fuchs-Tarlovsky V, Levin RM, Voss AC, Piemonte T. Oncology Evidence-Based Nutrition Practice Guideline for Adults. Journal of the Academy of Nutrition and Dietetics. 2017;117(2):297-310.e47. doi:10.1016/j.jand.2016.05.010
- Dignass AU, Gasche C, Bettenworth D, et al. European consensus on the diagnosis and management of iron deficiency and anaemia in inflammatory bowel diseases. Journal of Crohn's & colitis. 2015;9(3):211-22. doi:10.1093/ecco-jcc/jju009
- Rubio-Tapia A, Hill ID, Kelly CP, Calderwood AH, Murray JA. ACG clinical guidelines: diagnosis and management of celiac disease. The American journal of gastroenterology. 2013;108(5):656-76; quiz 677. doi:10.1038/ajg.2013.79
- Rubio-Tapia A, Hill ID, Semrad C, et al. American College of Gastroenterology Guidelines Update: Diagnosis and Management of Celiac Disease. Official journal of the American College of Gastroenterology | ACG. 2023;118(1):59-76. doi:10.14309/ajg.0000000000002075
- Devalia V, Hamilton MS, Molloy AM. Guidelines for the diagnosis and treatment of cobalamin and folate disorders. Br J Haematol. 2014;166(4):496-513. doi:10.1111/bjh.12959
- Killick SB, Bown N, Cavenagh J, et al. Guidelines for the diagnosis and management of adult aplastic anaemia. Br J Haematol. 2016;172(2):187-207. doi:10.1111/bjh.13853
- BCSH. Guidelines for the Investigation and Management of Vitamin B12 and Folate Deficiency. Updated April, 2018. https://www.hey.nhs.uk/wp/wp-content/uploads/2016/03/vitaminB12FolateDeficiency.pdf
- Mikhail A, Brown C, Williams JA, et al. Renal association clinical practice guideline on Anaemia of Chronic Kidney Disease. BMC nephrology. 2017;18(1):345. doi:10.1186/s12882-017-0688-1
- NCCN. NCCN Guidelines Myelodysplastic Syndromes. Updated January 12, 2026. https://www.nccn.org/professionals/physician_gls/pdf/mds.pdf
Coding Section
| Code | Number | Description |
| CPT | 82746 | Folic acid; serum |
| 82747 | Folic acid; RBC | |
| 0399U | Neurology (cerebral folate deficiency), serum, detection of anti-human folate receptor IgG-binding antibody and blocking autoantibodies by enzyme-linked immunoassay (ELISA), qualitative, and blocking autoantibodies, using a functional blocking assay for IgG or IgM, quantitative, reported as positive or not detected | |
| ICD-10 Diagnoses Codes | D53.1 | Other Megaloblastic Anemias, (NEC) |
| D53.9 | Nutritional anemia, unspecified | |
| D52.0 | Dietary folate deficiency anemia | |
| D52.1 | Drug-induced Folate Deficiency Anemia | |
| D52.8 | Other Folate Deficiency Anemias | |
| D52.9 | Folate Deficiency Anemia, Unspecified; Folate Deficiency Anemia, NOS | |
| D75.89 | Other specified diseases of blood and blood-forming organs | |
| E53.8 | Deficiency of other specified B group vitamins | |
| Z79.899 | Other long term (current) drug therapy |
Procedure and diagnosis codes on Medical Policy documents are included only as a general reference tool for each policy. They may not be all-inclusive.
This medical policy was developed through consideration of peer-reviewed medical literature generally recognized by the relevant medical community, U.S. FDA approval status, nationally accepted standards of medical practice and accepted standards of medical practice in this community, and other nonaffiliated technology evaluation centers, reference to federal regulations, other plan medical policies, and accredited national guidelines.
"Current Procedural Terminology © American Medical Association. All Rights Reserved"
History From 2018 Forward
| 05/05/2026 | Annual review, no change to policy intent. Updating table of terminology, rationale, and references. |
| 07/23/2025 | Annual review, updating criteria to allow serum folic acid testing for all types of anemia and for those who will or who have already undergone bariatric procedures. Also updating description, rationale, and references. |
| 10/03/2024 | Annual review, no change to policy intent. Updating rationale and references. |
| 07/29/2024 | Changing the Review date to 10/01/2024. |
| 07/26/2023 | Annual review, adding policy statement regarding folate receptor antibody testing, updating criteria for folate testing to require member is diagnosed with megaloblastic or macrocytic anemia and has been treated with folate prior to testing. Also updating description, table of terminology, rational, and references. Add PLA CODE 0399u |
| 06/05/2023 | Adding Code 0399U to coding section. No other changes made to policy. |
| 07/26/2022 | Annual review, no change to policy intent, adding definition of RBC in criteria #3. Also updating description, rationale and references. |
| 07/09/2021 |
Annual review, no change to policy intent. Updating rationale and references. |
| 07/16/2020 |
Annual review, no change to policy intent. Updating background, guidelines and references. |
| 07/16/2019 |
Updated policy to include diagnosis of megaloblastic or macrocytic anemia. No other changes made. |
| 07/12/2019 |
Annual review, no change to policy intent. |
| 10/30/2018 |
Updating review date. No other changes made. |
| 06/26/2018 |
New Policy |